by
K.L. Heong, International Rice Research Institute, Los Baños, Philippines

Insecticides in SE Asia are sold like FMCGs in village level retailer shops that are usually not properly licensed.
Rice planthopper outbreaks are insecticide-induced. Research at IRRI and elsewhere have all clearly demonstrated these impacts. Details are summarized in Way and Heong (1994), Heong and Schoenly (1998), Heong (2009) and Bottrell and Schoenly (2012). All insecticides are biocides acting as neurotoxins interfering with normal physiological functions and thus all animals (including humans) are susceptible to them. Natural enemies in the rice ecosystem tend to be more vulnerable because they move about to forage and many live in the water where most of pesticides sprayed eventually add up. Insecticide sprays thus disproportionately kill more natural enemies. Among the insecticide used, some active ingredients are more hazardous to the rice ecosystem. A procedure was developed by Kovach et al (1992) to measure the environmental impact using environmental impact quotient (EIQ) values that are maintained and up dated by Cornell University. The table of EIQs may be obtained from http://www.nysipm.cornell.edu/publications/eiq/files/EIQ_values_2012insect.pdf
Brown planthopper outbreaks are beginning to occur in several places in Indonesia (see links below) and Thailand in the last few months. There are shifts in active ingredients being marketed in many Asian countries. Some of the pyrethroids are gradually been replaced by neonicotinoids that are being phased out in Europe. Some of these have recently been shown to be highly toxic to the aquatic fauna.
In assessing the potential of an insecticide in causing BPH outbreaks, 5 main categories of the ecological properties were scored and the scoring scheme is shown in Table 1.
Table 1: Scoring scheme of variables that can potential cause BPH outbreaks
| Variables | Range of values | Scores |
| Toxicity to bees (and parasitoids) | >100mu g, 2 – 100 mu g, <2 mu g | 1, 3, 5 |
| Toxicity to fish (and aquatic predators) | > 10 ppm , 1-10 ppm, < 1 ppm | 1, 3, 5 |
| Toxicity to beneficials (and spiders) | Low, Moderate, Severe | 1, 3, 5 |
| Systemic in action | Non systemic, systemic | 1,3 |
| Residual effects (leaf surface half life (days) | l-2 weeks, 2-4 weeks, > 4 weeks | 1, 3, 5 |
High toxicity to bees which are hymenopterans would indicate that the insecticide would have high toxicity to all hymenopteran parasitoids. Similarly high toxicity to fish indicates high toxicity to the aquatic fauna.
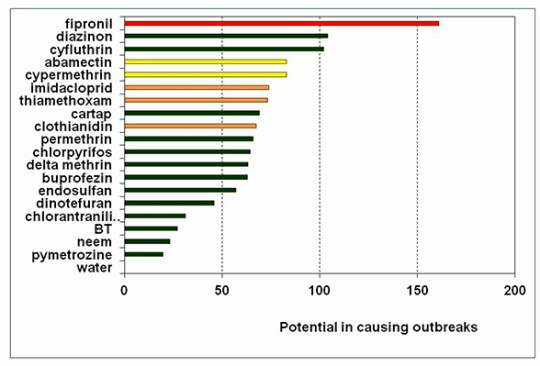
Potential of causing BPH outbreaks in some insecticides used in rice fields. The higher values indicate that these insecticides have higher potential to cause BPH outbreaks when used in rice fields.
The scores of all the 5 categories are cummulated and compared with water, which is expected to have a zero potential of causing a BPH outbreak. All insecticides have the potential of causing BPH outbreaks with fipronil the highest potential. This insecticide has been removed from the market in most parts of China and is being promoted in South East Asia. Similarly imidacloprid has also been phased out from the China market because of resistance development. In the SE Asian market however the trend is towards increasing the use of neonicotinoids as the carbamates, organophosphates and pyrethroids begin to phase out. Between 1997 and 2010 there was a shift to neonicotinoids and insecticide with other modes of actions in the world insecticide market (Casida and Durkin 2013).
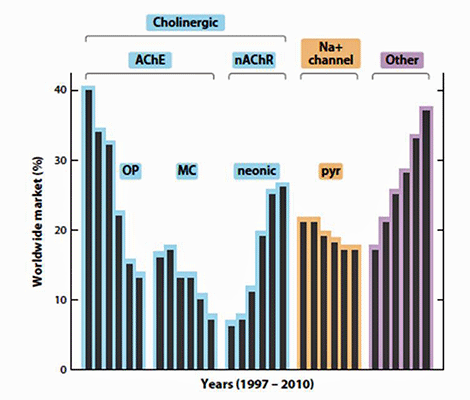
Changing trends in world insecticide use from OPs (organophosphates), MCs (methylcarbamates) and pyrethroids (Pyr) to neonics (neonicotinoids) and insecticides with other modes of action (From Casida and Durkin, 2013)
Although the neonicotinoids have slightly lower potentials in causing outbreaks than cypermethrin and abamectin, they can still potentially be threats, especially when market promotion will increase their use and misuses. In addition the neonicotinoids have much higher residual effects, sometimes referred to as the “new DDT”.
In China insecticide use in rice is shifting towards the use of chlorantraniliprole and pymethrozine both with low potentials in causing BPH outbreaks. Perhaps we might see less outbreaks in China in the near future if misuse is well managed and the development of insecticide resistance is controlled.
References
Bottrell, D.G. and Schoenly, K.G. 2012. Resurrecting the ghost of green revolutions past: the brown planthopper as a recurring threat to high-yielding rice production in tropical Asia. Journal of Asia-Pacific Entomology 15:122-140. (pdf)
Casida, J.E. and Durkin, K.A. 2013. Neuroactive insecticides: Targets, selectivity, resistance and secondary effects. Annu. Rev. Entomol., 58, 99 – 117. (pdf)

